

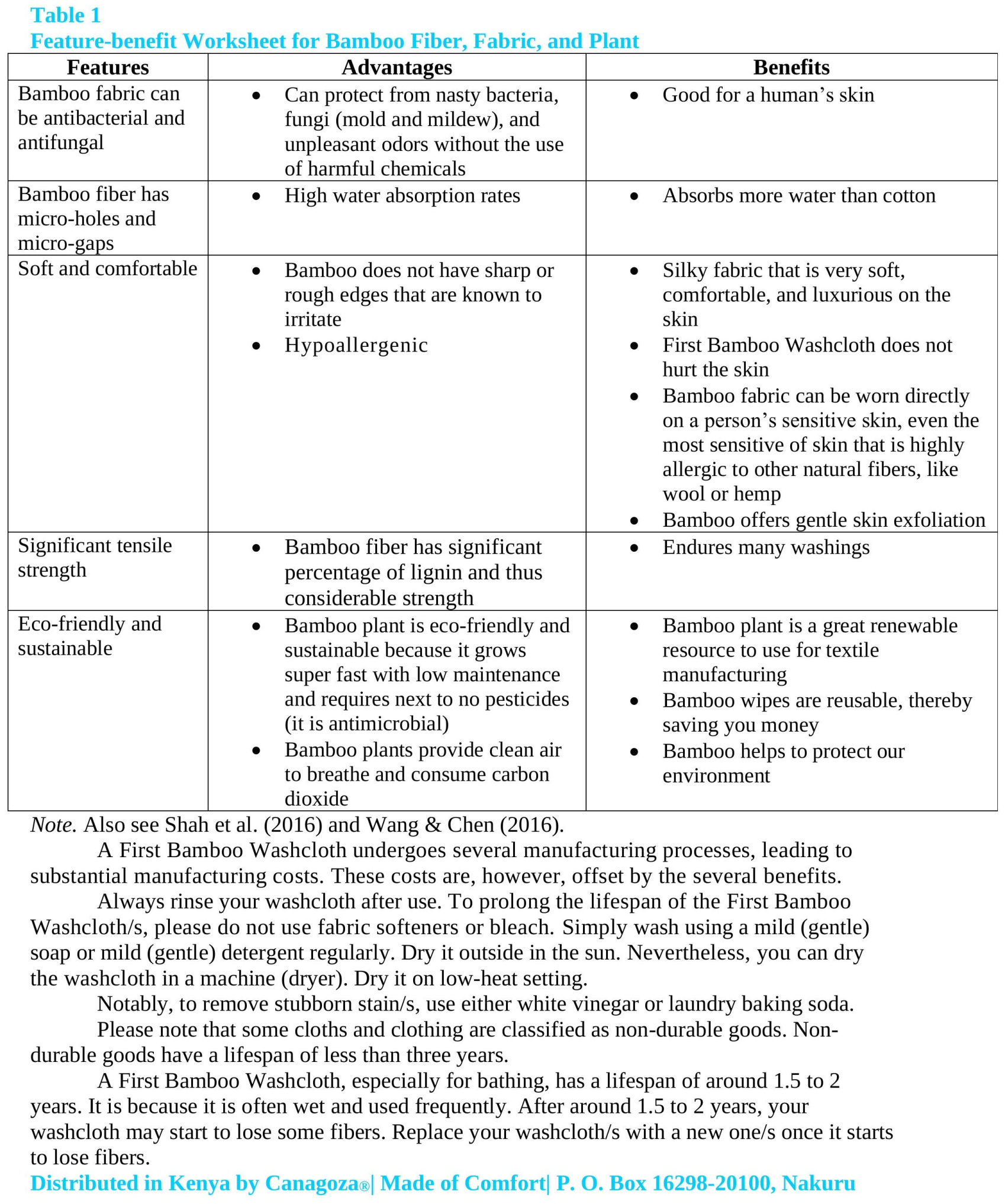
Collins English Dictionary defines a washcloth as a small cloth that you use for washing yourself. According to Merriam-Webster Dictionary, a washcloth is a cloth that is used for washing or bathing one's face and body. It is also called facecloth or washrag.
Notably, bamboo is often blended with cotton (in the ratio of 70% bamboo to 30% cotton) to give the fabric a little more structure (Wang & Chen, 2016).
Removing Soap Residue and Stubborn Stain/s
If the washcloth for bathing is coated with soap after rinsing/washing, hold it using one of your hands. Place your thumb and index finger of the other hand on the washcloth (the washcloth is now at the center).
Gently apply pressure on the washcloth. Move the thumb and index finger downward to remove the soap.
Removing Stubborn Stain/s
Add some warm water in a basin. Add white vinegar (as per manufacturer’s instructions). Place the washcloth/s. Allow the washcloth to soak for some time (perhaps overnight).
Next, wash it in plain water (no detergent, no fabric softener/conditioner...). Rinse thoroughly and dry, or just use it when wet for bathing (Abgarian, 2020; Leverette, 2021a; “White vs. distilled vinegar,” n.d.).
Alternatively, to remove stubborn stain/s, dissolve some laundry baking soda in some warm water. Add some cool water in a basin (or other container). Add the dissolved baking soda. Next, place the washcloth.
Allow the washcloth to soak for a significant timeframe (probably overnight). Next, wash it in plain water (no detergent, no fabric softener/conditioner...). Rinse thoroughly and dry, or just use it when wet for bathing (Arm & Hammer, 2019; Leverette, 2021b).
Can I use Baking Soda and White Vinegar Together?
No, when used together, baking soda and white vinegar will neutralize one another (Tide, 2020).
Mild (Gentle) Soaps Description
Some soaps are not “true” soaps. True soap is a combination of natural fats and an alkali (lye). Lye is also known as sodium hydroxide (Higuera, 2019). Sodium hydroxide is a chemical that comes from salt.
Today, however, most ordinary or traditional soaps do not contain lye or natural fat. These traditional soaps are actually synthetic detergents or cleansers. They can contain fragrance, sodium lauryl sulfate, and other ingredients that are harsh to your skin (Higuera, 2019). The average pH level in traditional soap is 9 to 10. However, your skin’s normal pH level is usually 4.5 to 6.5 (Siddons, 2019).
Traditional soaps can alter the pH balance (acidity level) of your skin by increasing skin pH (Siddons, 2019). The alkaline pH of the traditional soaps can elevate skin pH above 8.0 and destroy the skin lipid layer (Mendes et al., 2016). It can lead to skin dryness, acne itching, and irritation.
By contrast, mild (gentle) soap does not affect the pH of your skin (Higuera, 2019; Siddons, 2019).
References
Please click on (or tap on) the following link to access/view the "References": References -- First Bamboo Washcloths.doc